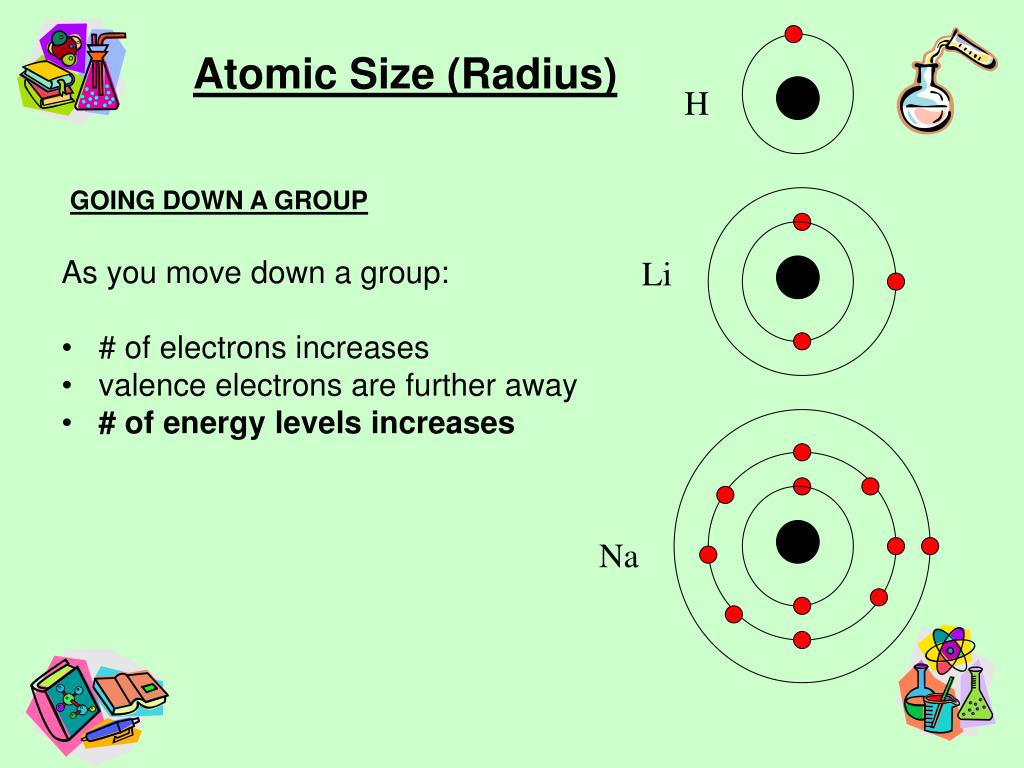
Therefore, the first ionization energy ( #"IE"_1#) for, say, #"B"#, is lower than for #"Be"#, because the #2p# orbitals are higher in energy than the #2s#, and so, #"B"# can be more easily ionized than #"Be"#.įor elements with electrons in a subshell of #l >= 1#, once electron pairing starts occurring, the paired electron is easier to remove than an unpaired electron.Įlectron pairing causes electron repulsion between like-charges which means that the ionization energy required to remove the first electron ( #"IE"_1#) is smaller. The new subshell is higher in energy (less core-like). There are, however, some exceptions to this general rule, as you can see in the diagram below:Įlements on a new row have their final electron start on a new quantum level, so naturally its ionization energy drops a bunch.Įlements in the same row that start to fill a new subshell ( a new #bb(l)#) have a drop in ionization energy downwards. So, in general, ionization energy increases from the bottom-left to the upper-right of the periodic table. The smaller the atomic radius, the more closely the electrons are held by the nucleus, and thus the higher the ionization energy. Note that it means you cannot, say, compare #"N"# and #"S"# fairly, since they have conflicting periodic trends.Īs I said, the ionization energies follow somewhat from atomic radius. This new quantum level is farther out, and therefore, the radius increases as we go downwards on the periodic table. 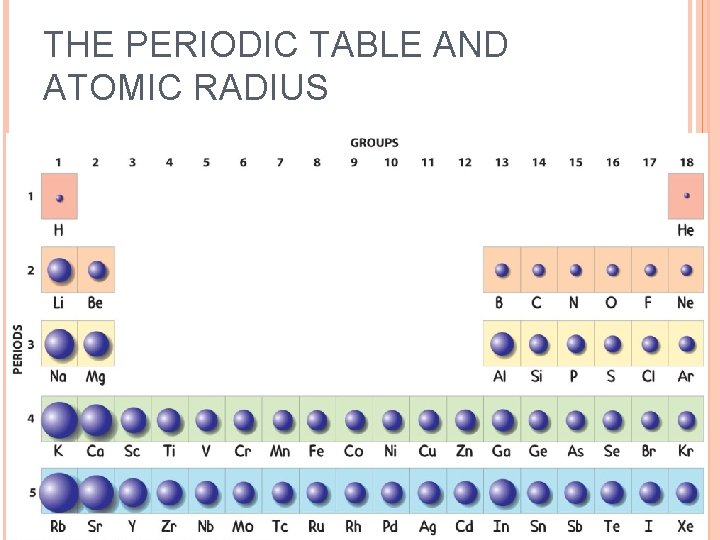
Higher #Z_"eff"# means smaller radius.Ī new quantum level corresponds to the valence shell on a new row (below). In general, the atomic radius (except for many transition metals) has a pattern where it decreases from the bottom-left to the top-right of the periodic table.Įffective nuclear charge increases from left to right.Īs we go across the periodic table from left to right, protons are more massive than electrons, so adding one proton and one electron to obtain a new atom means that #bb(Z_("eff")uarr)#. Ii) electrons with an equal principal quantum number and a smaller azimuthal quantum number l.Of these, atomic radius is the most predictable, and ionization energy and electron affinity trends follow (at least in part) from such trends. I) electrons with a smaller principal quantum number n and If the group is of the (d) or (f), type, an amount of 1.00 for each electron "closer" to the atom than the group. If the group is of the (s p) type, an amount of 0.85 from each electron with principal quantum number n one less than that of the group, and an amount of 1.00 for each electron with principal quantum number two or more less. The shielding constant for each group is formed as the sum of the following contributions:Īn amount of 0.35 from each other electron within the same group except for the (1s) group, where the other electron contributes only 0.30. The nucleus has its character positive charge, but due to shielding by core electrons the total positive charge is not completely felt by the electrons, so the actual net positive charge felt by the valence electron has its own name and is called the effective nuclear charge $Z_$ etc.Įach group is given a different shielding constant which depends upon the number and types of electrons in those groups preceding it. Valence electrons experience an electrostatic force from the nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
